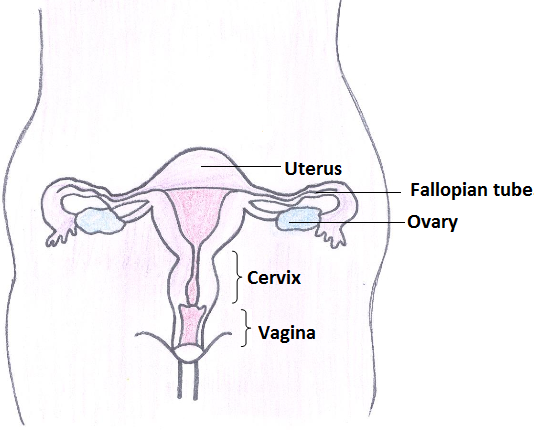
The cervix is the lower part of the womb also known as uterine cervix. The cervix connects the body of the uterus to the vagina(birth canal). The part of the cervix closest to the body of the uterus is called the endocervix. The part next to the vagina isthe ectocervix. It is about 2-3 cms in length.
Epidemiology
More women in India die from cervical cancer than in any other country.
Around 1.23 lakh new women are diagnosed with cervical cancer in India and 67,500 women die of cervical cancer in India every year (Globocan 2012) [1].
The incidence of cervical cancer is higher in rural areas when compared to cities [2].
Risk Factors [3]
- Persistent infection of the cervix with Human Papillomavirus (HPV)
- Giving birth to many children
- Having many sexual partners
- Spouse having multiple sexual partners
- Having first sexual intercourse at a young age
- Smoking
- Due to HIV/AIDS, immunosuppressive drugs, transplant etc.
The presence of these risk factors does not imply that you will definitely acquire cancer. However, if you have one or more of these risk factors, it is advisable to consult your doctor.
Prevention
(I) Primary Prevention: It is designed to prevent the disease from occurring in the first place.
- Adopt safe sex practices (avoid multiple sexual partners).
- Use of male condoms as barrier contraceptives to reduce the risk of HPV infection.
- Timely treatment of reproductive tract infections.
- There is evidence that circumcision for men may reduce the incidence of infection among sexual partners.
- HPV vaccination: Prophylactic vaccines for cervical cancer target HPV 16 and 18, the most common oncogenic types of HPV responsible for cervical cancer. HPV vaccination is not effective against all oncogenic HPV types. Currently two vaccines, licensed globally are available in India; a quadrivalent vaccine (against HPV genotypes 6, 11, 16, 18) and a bivalent vaccine (against HPV genotypes 16, 18). The vaccine dose is 0.5 ml given intramuscularly, either in the deltoid muscle or in the antero-lateral thigh. It is available as a sterile suspension for injection in a single-dose vial or a prefilled syringe. The recommended age for initiation of vaccination is 9–14 years. Catch-up vaccination is permitted up to the age of 26 years.
Females who have not been exposed to the HPV infection are likely to benefit more from the vaccine.
Indian Academy of Pediatrics (IAP) recommendations on HPV vaccination[4]:
- Only 2 doses of either of the two HPV vaccines for adolescent/pre-adolescent girls aged 9-14 years
- For girls 15 years and older, and immune-compromised individuals 3 doses are recommended
- For two-dose schedule, the minimum interval between doses should be 6 months.
- For 3 dose schedule, the doses can be administered at 0, 1-2 (depending on brands) and 6 months
Vaccine does not guarantee complete protection against cervical cancer. Cervical screening tests are still important as:
- Immunization with the HPV vaccine will take several years to reduce the chances of developing cervical cancer.
- The vaccine does not protect against all HPV types.
- Not all cases of cervical cancer are caused by the high-risk HPV16 and HPV18 strains.
Duration of protection of vaccine:
The duration of vaccine protection is unclear. Current studies indicated that the vaccine is effective for at least 5 years. Ongoing studies are investigating the long term efficacy of the vaccine.
VACCINATION IS NOT A REPLACEMENT FOR CERVICAL CANCER SCREENING
Currently, there are no guidelines on HPV vaccination in our country.
(II) Secondary Prevention: Secondary prevention aims at detecting the disease in its early stages (pre-cancers) through screening and to prevent its progression.
Screening tests are done in apparently healthy women to diagnose changes in the cervix which are pre-cancerous and could develop into cervical cancer in future. If the abnormal tissue or cells are removed, the disease can be prevented from progressing to cancer [5]. Available screening tests for cervical cancer include Pap smear test, VIA (visual inspection with acetic acid), VILI (visual inspection with Lugol’s iodine) and HPV DNA test.
The Pap (Papanicolaou) test is a method that has been used for over 60 years to detect potentially pre-cancerous and cancerous changes in cells of the cervix.
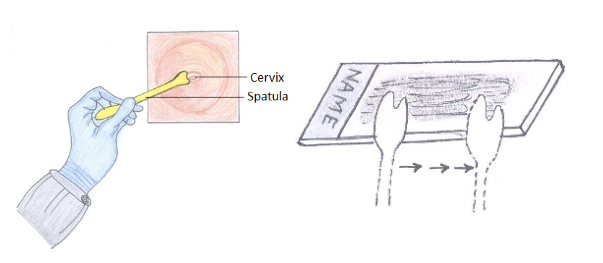
Pap smear is a simple and painless test. An instrument called a speculum is introduced into the vagina to visualize the cervix. The health care provider will then use a special stick or brush to take a few cells from the surface of and inside the cervix. The cells are placed on a glass slide and sent to a lab for cytologic testing. The Pap test is done in married women above 21 years and should be repeated once in every 3 years. If this test is combined with HPV test (for women above 30 years of age), then the duration of screening can be increased to 5 years.
Pap Test
Other tests used for screening of cervical cancer
HPV test is a laboratory test used to check the presence of DNA or RNA for certain types of HPV. Cells are collected from the cervix and are checked to find out if an infection is caused by a type of HPV that is linked to cervical cancer [7]. This test may be done using the sample of cells removed during a Pap test. This test is not recommended in women aged below 30 years as HPV infection is very common in this age and may be spontaneously cleared within 2 years.
Visual inspection of the cervix, using acetic acid (VIA) or Lugol’s iodine (VILI) is also used as a screening test to find precancerous lesions[8]. Such procedures do not need laboratories and transport of specimens, require very little equipment and provide women with immediate test results. A range of medical professionals-doctors, nurses, or professional midwives-can effectively perform the procedure, provided they receive adequate training and supervision.
Abnormal Pap tests
Abnormal Pap test results usually do not mean you will always have cancer. Most often there is a small problem with the cervix. If results of the Pap test are unclear or show a small change in the cells of the cervix, your doctor may repeat the Pap test immediately, in 6 months or a year, or run more tests. Some abnormal cells will turn into cancer. Treating abnormal cells that don’t go away on their own can prevent almost all cases of cervical cancer. If you have abnormal results, talk to your doctor about what they mean. If treatment is needed, it can be done early enough to prevent cancer before it develops. Treatment is often done in an out patient department (OPD). If the test finds more serious changes in the cells of the cervix, the doctor will suggest more tests. Results of these tests will help your doctor decide on the best treatment.
The Pap smear is a simple test to collect a small sample of cells from the cervix which helps to diagnose precancerous and cancerous conditions of the cervix. It also aids in diagnosing infections and inflammation of the lower reproductive tract.
Who should get the Pap test done?
As per the International recommendations, the age to initiate screening is 21 years.
Women who are 30 years and above should undergo a Pap test once in every 3 years until the age of 65 years. If this test is combined with HPV test, then the duration of screening can be increased to 5 years.
Women who do not routinely require Pap test
- Women aged below 21 years and above 65 years
- Women who have undergone hysterectomy for benign condition
When should the Pap test be done?
The Pap test yields optimum results if scheduled between 10 to 20 days from the first day of menstrual period. The woman should not be menstruating at the time of test.
Preparation for Pap smear
Following should be avoided 48 hours before the test:
- Intercourse
- Douching of vagina
- Vaginal medications
- Vaginal contraceptives like creams/ jellies
Procedure
An instrument called a speculum is gently introduced into the vagina to visualize the cervix. There may be some discomfort or cramping during the procedure, but it is not usually painful.
- A small wooden stick or spatula is used to gently scrape the surface of the lower part of the cervix to pick up cells.
- A special brush, called a cytobrush is used to obtain cells from the inner part of the cervix.
- The cells are placed on a glass slide, immediately fixed in ethanol and sent to laboratory for further processing and interpretation.
Results of Pap test
A Pap test result may be reported as normal or abnormal.
- Normal Pap test
If the test report is normal, this means no abnormal or cancerous cells have been found in the smear taken.
- Abnormal Pap tests
Abnormal Pap test results usually do not mean that the woman has cancer. Most often there is a small problem with the cervix. If results of the Pap test are unclear or show a mild abnormality in the cells of the cervix, your doctor may repeat the Pap test in 6 weeks, in 6 months or a year, or run more tests. Treating abnormal cells that don’t go away on their own can prevent almost all cases of cervical cancer. Treatment of this abnormality is often done in an out-patient department (OPD). If the test findings suggest more severe abnormality in the cells, it is confirmed by further diagnostic procedures:
Colposcopy: A procedure in which a colposcope (a lighted, magnifying instrument) is used to check the vagina and cervix for abnormal areas.
Biopsy: A sample of tissue is cut from the cervix and viewed under a microscope by a pathologist to check for signs of cancer. A biopsy that removes only a small amount of tissue (punch biopsy) is usually done in the OPD.
Signs and Symptoms
- Abnormal vaginal bleeding: Bleeding and spotting between periods, unusually longer or heavier periods, bleeding after menopause
- Unusual or excessive vaginal discharge with foul smell
- Vaginal bleeding after having sexual intercourse
- Pain in the lower abdomen or pelvic pain
- Pain during sexual intercourse
The presence of these signs and symptoms does not imply that you are suffering from cancer. However, if you have one or more of these, it is advisable to consult your doctor.
Diagnosis
If any of the screening tests (Pap test, VIA, HPV test) are found to be positive, further testing may be necessary to determine whether the changes are cancerous. A colposcopy may be performed and/or a small sample of tissue (biopsy) will be obtained from the cervix
- Colposcopy: A procedure in which a colposcope (a lighted, magnifying instrument) is used to check the vagina and cervix for abnormal areas.
- Biopsy: If abnormal cells are found in a Pap test, the doctor may do a biopsy. A sample of tissue is cut from the cervix and viewed under a microscope by a pathologist to check for signs of cancer. A biopsy that removes only a small amount of tissue is usually done in the out patient department (OPD). A woman may need to go to a hospital for a cervical cone biopsy (removal of a larger, cone-shaped sample of cervical tissue).
taging [9]
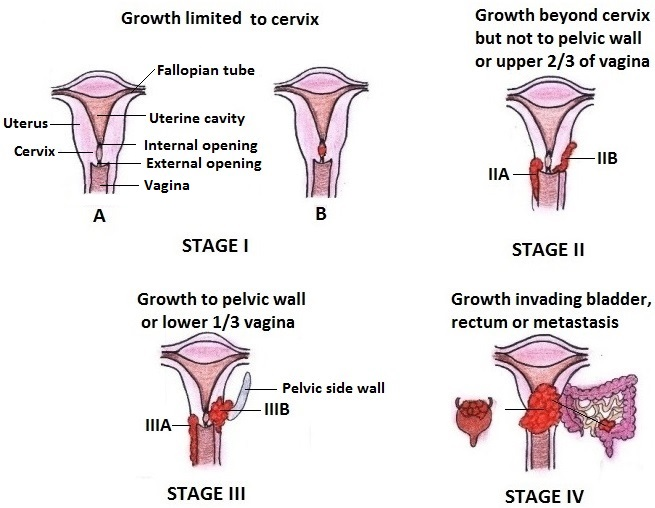
The process to find out the extent of disease is called staging.
Stage 0 or Carcinoma in situ: abnormal cells are found in the innermost lining of the cervix which may not be seen to naked eye.
Stage I: In this stage, cancer is limited to cervix only.
Stage II: Cancer has spread beyond the cervix but not to the tissues that line the part of the body between the pelvic wall or to the lower third of the vagina.
Stage III: Cancer has spread to the lower third of the vagina, and/or to the pelvic wall, and/or has caused kidney problems.
Stage IV: Cancer has spread to the bladder, rectum, or other parts of the body.
Treatment
Three types of treatment are used to treat cervical cancer viz. surgery, radiotherapy and chemotherapy. These therapies may be given alone or in combination with one another. Treatment depends on the stage of the cancer, the type of tumor cells and a woman’s medical condition.
Treatment of pre-cancerous lesions may include the following: Removal or destruction of the part of the cervix affected by disease.
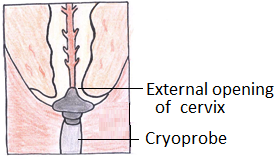
- Cryosurgery: application of freezing probe to destroy abnormal or diseased tissue in the cervix for about 5 minutes.
- Loop electrosurgical excision procedure (LEEP): procedure to remove abnormal and or cancerous cells in the cervix using a thin, low-voltage electrified wire loop that acts as a knife.
Treatment
Three types of treatment are used to treat cervical cancer viz. surgery, radiotherapy and chemotherapy. These therapies may be given alone or in combination with one another. Treatment depends on the stage of the cancer, the type of tumor cells and a woman’s medical condition.
Treatment of pre-cancerous lesions may include the following: Removal or destruction of the part of the cervix affected by disease.
- Cryosurgery: application of freezing probe to destroy abnormal or diseased tissue in the cervix for about 5 minutes.

- Loop electrosurgical excision procedure (LEEP): procedure to remove abnormal and or cancerous cells in the cervix using a thin, low-voltage electrified wire loop that acts as a knife.

- Laser surgery: Using a laser beam to burn of abnormal cells in the cervix.
- Conization: Excision of a cone-shaped sample of tissue from the mucous membrane of the cervix by using cold knife or scalpel. This procedure is carried out under general anesthesia in a hospital operating room.

- Hysterectomy: for women whose tumor cannot be completely removed by conization and who no longer want to have children.
Stage I Cervical Cancer
Treatment of stage I cervical cancer may include surgery, chemotherapy and/or radiation therapy depending on the sub-stage, age and desire of the patient and preference of the treating physician.
- Total hysterectomy with or without bilateral salpingo-oophorectomy
- Modified radical hysterectomy and removal of lymph nodes.
- Internal radiation therapy.
- Radical hysterectomy and removal of lymph nodes.
- Radical hysterectomy and removal of lymph nodes followed by radiation therapy plus chemotherapy.
- Radiation therapy plus chemotherapy.
- A combination of internal radiation therapy and external radiation therapy

Stage II Cervical Cancer
Treatment of stage II cervical cancer may include the following:
- Radical hysterectomy and removal of lymph nodes followed by radiation therapy plus chemotherapy.
- A combination of internal radiation therapy and external radiation therapy plus chemotherapy.
- Radical hysterectomy and removal of lymph nodes.
- Radical hysterectomy and removal of lymph nodes followed by radiation therapy plus chemotherapy.
Stage III Cervical Cancer
Treatment of stage III cervical cancer may include internal and external radiation therapy combined with chemotherapy
Stage IV Cervical Cancer
- Radiation therapy to relieve symptoms caused by the cancer and improves quality of life (radiation therapy as palliative therapy).
- Chemotherapy and targeted therapy (monoclonal antibodies). Chemotherapy as palliative therapy to relieve symptoms caused by the cancer and improve quality of life.
References
[1] Ferlay J, Soerjomataram I, Ervik M, et al. (2013) GLOBOCAN 2012 v1.0, Cancer Incidence and Mortality Worldwide: IARC Cancer Base No. 11.Lyon, France: http://globocan.iarc.fr. Accessed on 04 Sep 2014
[2] Kurkure AP, and Yeole BB. Social inequalities in cancer with special reference to South Asian countries. Asian Pac J Cancer Prev 2006;7:36-40
[3] Bosch FX, de Sanjosé S. Chapter 1: Human Papillomavirus and Cervical Cancer–Burden and Assessment of Causality. J Natl Cancer Inst Monogr 2003;31:3-13
[4] Vashishtha VM, Choudhury P, Kalra A, et al. Indian Academy of Pediatrics (IAP) recommended immunization schedule for children aged 0 through 18 years–India, 2014 and updates on immunization. Indian Pediatr. 2014 Oct; 51(10):785-800.
[5] Miller AB, Chamberlain J, Day NE, et al. Report on a workshop of the UICC project on evaluation of screening for cancer. Int J Cancer 1990;46:761-9
[6] Moyer VA; U.S. Preventive Services Task Force. Screening for cervical cancer: U.S. Preventive Services Task Force recommendation statement. Ann Intern Med 2012;156:880-91
[7] Sankaranarayanan R, Nene BM, Shastri SS, et al. HPV Screening for Cervical Cancer in Rural India. N Engl J Med 2009;360:1385-94
[8] Sankaranarayanan R, Budukh AM and Rajkumar R. Effective screening programmes for cervical cancer in low- and middle-income developing countries. Bull World Health Organ 2001;79:954-62
[9] Shepherd JH. Cervical and vulva cancer: changes in FIGO definitions of staging. Br J Obstet Gynaecol 1996;103:405-6
